International olive council: spectrophotometric investigation in the ultraviolet foreword
The method describes the procedure for performing a spectrophotometric examination of olive oil in the ultraviolet region
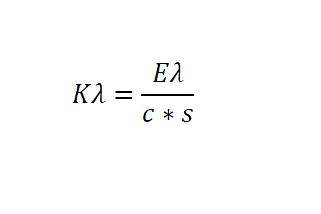
Spectrophotometric examination in the ultraviolet can provide information on the quality of a fat, its state of preservation and changes brought about by technological processes.
The absorption at the wavelengths specified in the method is due to the presence of conjugated diene and triene systems resulting from oxidation processes and/or refining practices. These absorptions are expressed as specific extinctions E1% (the extinction of 1% w/v solution of the fat in the specified solvent, in a 10 mm cell) conventionally indicated by K (also referred to as “extinction coefficient”).
1. SCOPE
The method describes the procedure for performing a spectrophotometric examination of olive oil in the ultraviolet region.
2. PRINCIPLE OF THE METHOD
A sample is dissolved in the required solvent and the absorbance of the solution is measured at the specified wavelengths with reference to pure solvent.
The specific extinctions at 232 nm and 268 nm in iso-octane or 232 nm and 270 nm in cyclohexane are calculated for a concentration of 1% w/v in a 10 mm cell.
3. EQUIPMENT
3.1. A spectrophotometer suitable for measurements at ultraviolet wavelengths (220 nm to 360 nm), with the capability of reading individual nanometric units. A regular check is recommended for the accuracy and reproducibility of the absorbance and wavelength scales as well as for stray light.
3.1.1. Wavelength scale: This may be checked using a reference material consisting of an optical glass filter containing holmium oxide or a holmium oxide solution (sealed or not) that has distinct absorption bands. The reference materials are designed for the verification and calibration of the wavelength scales of visible and ultraviolet spectrophotometers having nominal spectral bandwidths of 5 nm or less. The measurements are carried out against an air blank over the wavelength range of 640 to 240 nm, according to the instructions enclosed with the reference materials. A baseline correction is performed with an empty beam path at every slit width alteration.
The wavelengths of the standard are listed in the certificate of the reference material.
3.1.2. Absorbance scale: This may be checked using commercially available sealed reference materials consisting of acidic potassium dichromate solutions, in certain concentrations and certified values of absorbance at its λmax (of 4 solutions of potassium dichromate in perchloric acid sealed in four UV quartz cells to measure the linearity and photometric accuracy reference in the UV). The potassium dichromate solutions are measured against a blank of the acid used, after baseline correction, according to the instructions enclosed with the reference material. The absorbance values are listed in the certificate of the reference material.
Another possibility in order to check the response of the photocell and the photomultiplier is to proceed as follows: weigh 0.2000 g of pure potassium chromate for spectrophotometry and dissolve in 0.05N potassium hydroxide solution in a 1000 ml graduated flask and make up to the mark. Take precisely 25 ml of the solution obtained, transfer to a 500 ml graduated flask and dilute up to the mark using the same potassium hydroxide solution.
Measure the extinction of the solution so obtained at 275 nm, using the potassium hydroxide solution as a reference. The extinction measured using a 1 cm cuvette should be 0.200 ± 0.005.
3.2. Rectangular quartz cuvettes, with covers, suitable for measurements at the ultraviolet wavelengths (220 to 360 nm) having an optical path-length of 10 mm. When filled with water or other suitable solvent the cuvettes should not show differences between them of more than 0.01 extinction units.
3.3. One- mark volumetric flasks, capacity 25 ml, class A.
3.4. Analytical balance, capable of being read to the nearest 0.0001 g
4. REAGENTS
During the analysis, unless otherwise stated, use only reagents of recognized analytical grade and distilled or demineralized water or water of equivalent purity.
Solvent: Iso-octane (2,2,4 trimethylpentane) for the measurements at 232 nm and 268 nm and cyclohexane for the measurements at 232 nm and 270 nm, having an absorbance less than 0.12 at 232 nm and less than 0.05 at 270 nm against distilled water, measured in a 10 mm cell.
5. PROCEDURE
5.1. The sample must be perfectly homogeneous and without suspended impurities. If not, it must be filtered through paper at a temperature of approximately 30°C.
5.2. Weigh accurately approximately 0.25 g (to the nearest 1 mg) of the sample so prepared into a 25 ml graduated flask, make up to the mark with the specified solvent and homogenize (1)1. The resulting solution must be perfectly clear. If opalescence or turbidity is present, filter quickly through paper.
5.3. If necessary, correct the baseline (220-290 nm) with solvent in both quartz cells (sample and reference), then fill the sample quartz cell with the test solution and measure the extinctions at 232, 268 or 270 nm against the solvent used as a reference.
The extinction values recorded must lie within the range 0.1 to 0.8 or within the range of linearity of the spectrophotometer which should be verified. If not, the measurements must be repeated using more concentrated or more dilute solutions as appropriate.
1 Generally, a mass of 0.25 -0.30 g is sufficient for absorbance measurements of virgin and extra virgin olive oils at 268 nm and 270 nm. For measurements at 232 nm, 0.05 g of sample are usually required, so two distinct solutions are usually prepared. For absorbance measurements of olive pomace oils, refined olive oils and adulterated olive oils, a smaller portion of sample, e.g. 0.1 g is usually needed due to their higher absorbance.
5.4. After measuring the absorbance at 268 or 270 nm, measure the absorbance at λmax, λmax+4 and λmax-4. These absorbance values are used to determine the variation in the specific extinction (ΔK).
NOTE: λmax is considered to be 268 nm for isooctane used as solvent and 270 nm for cyclohexane.
6. EXPRESSION OF THE RESULTS
6.1. Record the specific extinctions (extinction coefficients) at the various wavelengths calculated as follows:
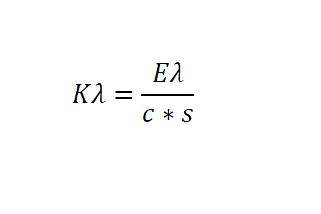
where:
K_ = specific extinction at wavelength ;
E_ = extinction measured at wavelength ;
c = concentration of the solution in g/100 ml;
s = path length of the quartz cell in cm;
expressed to two decimal places.
6.2. Variation of the specific extinction (ΔK)
The variation of the absolute value of the extinction (ΔK) is given by:
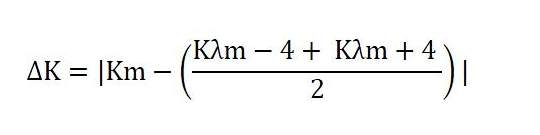
where Km is the specific extinction at the wavelength for maximum absorption at 270 nm and 268nm depending on the solvent used.
expressed to two decimal places.
COI/T.20/Doc. No 19/Rev. 3
February 2015
To comment you have to register
If you're already registered you can click here to access your account
or click here to create a new account


Comment this news